
- English
- Español
- Português
- русский
- Français
- 日本語
- Deutsch
- tiếng Việt
- Italiano
- Nederlands
- ภาษาไทย
- Polski
- 한국어
- Svenska
- magyar
- Malay
- বাংলা ভাষার
- Dansk
- Suomi
- हिन्दी
- Pilipino
- Türkçe
- Gaeilge
- العربية
- Indonesia
- Norsk
- تمل
- český
- ελληνικά
- український
- Javanese
- فارسی
- தமிழ்
- తెలుగు
- नेपाली
- Burmese
- български
- ລາວ
- Latine
- Қазақша
- Euskal
- Azərbaycan
- Slovenský jazyk
- Македонски
- Lietuvos
- Eesti Keel
- Română
- Slovenski
- मराठी
- Srpski језик
Development Trend of China's Pharmaceutical Intermediate Industry
2023-07-28
The rapid development of China's pharmaceutical intermediate industry is closely related to its national policies. Since 2016, China has issued clear documents to encourage small and medium-sized enterprises to develop new drugs, including chemical raw materials. At the same time, the structural upgrading of the pharmaceutical industry in developed countries has promoted the transfer of global related industrial chains and promoted China's chemical raw material drug supply chain. In addition, the increase in Chinese radiography technology has boosted product demand.
1. Policies stimulate the rapid development of the domestic pharmaceutical intermediate industry.
The pharmaceutical industry is related to the national economy and the people's livelihood. The entire upstream and downstream are involved in all aspects of the national economy. The Chinese government has long been directly or indirectly inclined to this policy. On March 16, 2016, the Fourth Session of the Twelfth National People's Congress voted and passed the resolution on the "Outline of the 13th Five-Year Plan for National Economic and Social Development." The document clearly encourages the research and development of new chemical drugs, including Chemical raw materials.
In 2016, in order to promote the innovative development, transformation and upgrading of the pharmaceutical industry, China compiled the "Guiding Opinions on Promoting the Healthy Development of the Pharmaceutical Industry", which put forward requirements for all aspects of the medical industry and specifically proposed support for the field of chemical raw materials.
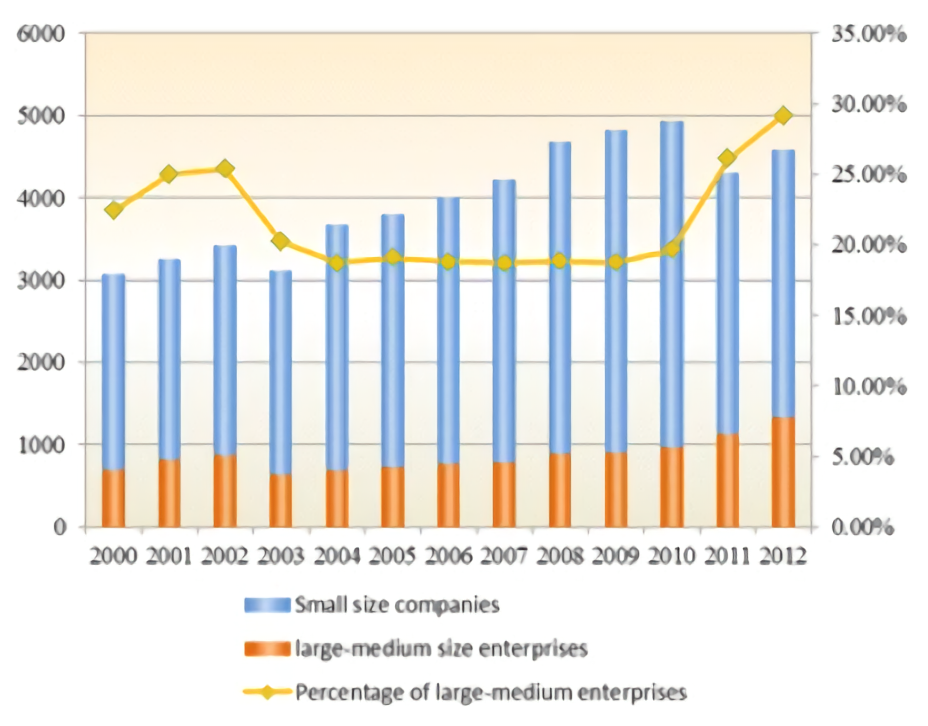
The number of pharmaceutical manufacture enterprises and percentage of large-medium enterprises in China.
Sourse: Obstacles and opportunities in Chinese pharmaceutical innovation
At the policy level, the emphasis on the medical and pharmaceutical industries and the direct support to the chemical raw material medicine industry have created a good political environment for the development of related enterprises and laid a foundation for the rapid development of the chemical raw material medicine industry. The good development prospects of the chemical raw material medicine industry will be directly passed to the upstream raw material industry, which will help stimulate the demand for the pharmaceutical intermediates market.
2. The shift in the global pharmaceutical industry chain increases the demand for raw materials.
With the structural upgrading of the pharmaceutical industry in developed countries, the transfer of related industrial chains has occurred globally, which has also had a profound impact on Chinese pharmaceutical companies.
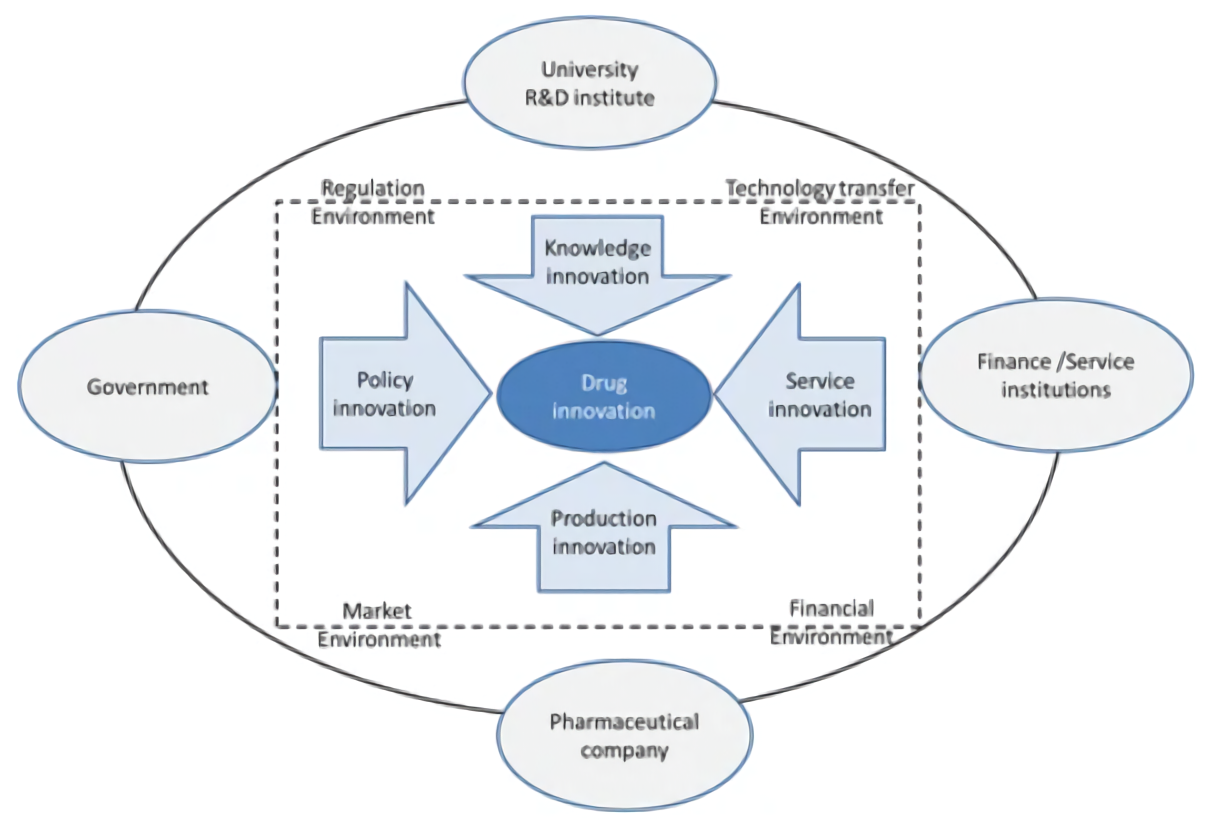
Pharmaceutical innovation system
Sourse: Obstacles and opportunities in Chinese pharmaceutical innovation
In recent years, large international pharmaceutical companies have gradually focused on the R&D and sales of patented drugs, and have outsourced traditional products level by level. Most of the contractors are pharmaceutical companies in emerging countries. Among them, in terms of chemical raw materials, my country occupies a large share in the bulk of raw materials, and has become one of the world's largest suppliers of chemical raw materials. At the same time, through years of research and advanced technology learning, more and more domestic companies have participated in the field of characteristic APIs with strong originality.
3. The increase of Chinese radiography equipment increases product demand
The use of contrast agent is different from other drugs, it needs to be used in conjunction with corresponding equipment. In recent years, China is in the middle of the growth of imaging equipment.
In 2004, China formulated and issued the "Administrative Measures for the Configuration and Use of Large-scale Medical Equipment." This approach regards X-ray computer tomography (CT) and medical magnetic resonance imaging equipment, and digital subtraction angiography X-ray machines with a value of 800 mA or more as the management products of Class B large-scale medical equipment. Purchase and use need to be reported to the provincial health administrative department for approval before approval.
However, with the comprehensive coverage of medical insurance in recent years, such basic medical facilities are becoming widespread, and the number of equipment has been directly linked to the number of patients in the relevant implementation rules. Therefore, as the demand for contrast equipment continues to increase, it will have a greater boost to the upstream contrast agent and its raw material market.




